REFINING AND BIOREFINING
In today’s world, the term refining is immediately reminiscent of oil and its tremendous development during the twentieth century that changed the life of humanity, mostly for the better, before threatening it today. by increasing the carbon dioxide content of the atmosphere.
Between apocalyptic forecasts and denials, there is a happy medium on which the progressive but obligatory development of renewable energies must be built. They are based almost exclusively on the sun, from direct photovoltaic or thermal capture to indirect via photosynthesis, which converts carbon dioxide and water naturally present in the atmosphere into food and non-food plant matter according to the following famous reaction scheme.

It is worth recalling at this point that both oil and coal are only biochemical and chemical conversions of dead plants and organisms at a time when the atmosphere was saturated with carbon dioxide. Their progressive fossilization has contributed to the increase of the oxygen content of the Earth’s atmosphere that we know today.
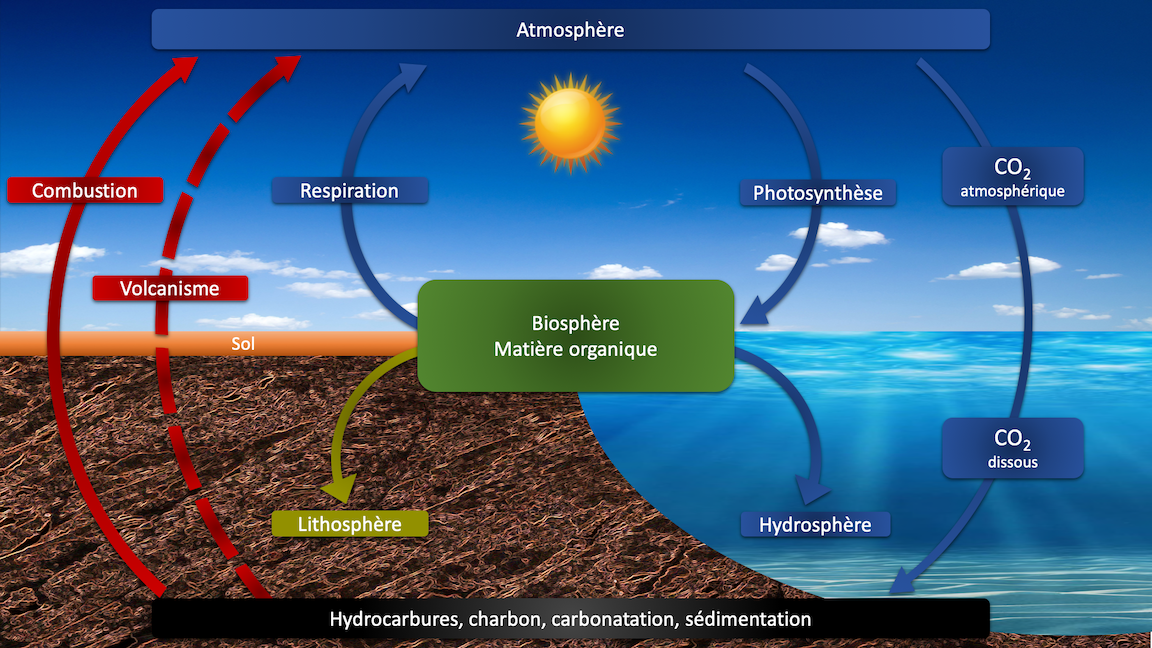
The reverse process underway since the beginning of the industrial age will certainly not bring the Earth’s atmosphere back to the early ages of the Earth but must stop because of a significant increase in the greenhouse effect with the induced climate changes that we let’s start feeling.
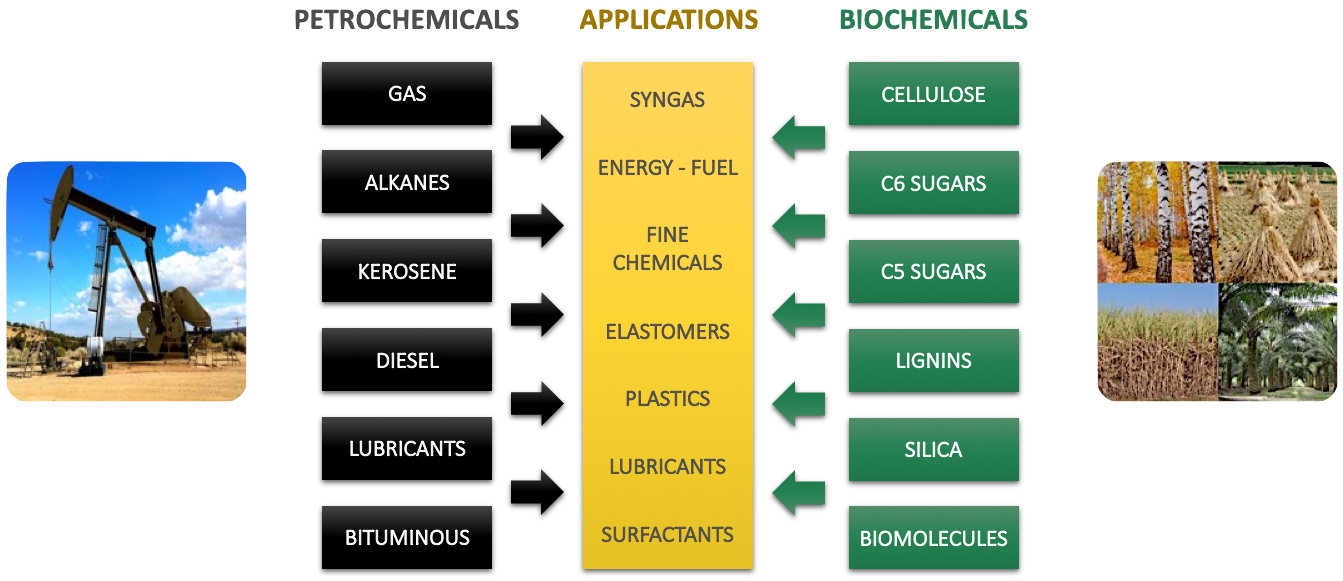
Since oil and coal are derived directly from non-food biomass, why not make the molecules that are today derived from petroleum and coal directly from this biomass?
This reasoning is simple and full of common sense since chemists and biochemists showed during the twentieth century that it was quite possible but with a serious downside.
Indeed, the inability to separate under acceptable economic and ecological conditions the components of biomass, as is commonly practiced with petroleum components, prohibited their development in full competitiveness with them.
It is not my purpose to recall the innumerable methods that have been studied for more than a century. if we take as historical reference the discovery of the separation of cellulose and the production of paper from wood in the late nineteenth century, they were and still are daily a long litany of failures.
Having for my part, taught for 25 years the genesis, extraction, refining and chemistry of petroleum at the University of Toulouse, I could appreciate the excellence of the petrochemical industry, when operations are conducted with rigor , and considered very quickly that only this model, applied to the transformation of non-food biomass, could allow its economic emergence for the greater good of our environment.
FROM OIL REFINERY TO LIGNOCELLULOSIC BIOMASS REFINERY
The initial distillation of crude oil which allows without degrading to separate the hydrocarbons it contains is inconceivable with wood, straw or bagasse.
If we want to go to the same molecules and materials, it will not be necessary to degrade, during the initial separation, their components that are cellulose, hemicelluloses, lignin and silica. The following diagram illustrates the purpose :
The first work carried out in my Laboratory at the University of Toulouse in the early nineties quickly showed me that the separation of the components of the lignocellulois biomass conducted in the water could only go towards the extraction cellulose at the cost of hard and polluting methods, never far from the current conditions of industrial production of chemical pulp
Sulfur used in various forms, in a very strongly basic or acidic medium, protects cellulose as well as possible to make pulp, but it will then be recycled what is anything but simple. Lignins and hemicelluloses are severely degraded to the point of having no choice but to incinerate them to recover energy to run the plants and recover the chemicals used as catalysts and reagents.
This is the situation that still prevails today for the only “biorefineries”, still a pulp or sugar factory, existing industrially from lignocellulosic biomass and whose concept and methods date back to the end of the 19th century.

This simply means that to produce 1 Ton of crude cellulose by these methods, 1 Ton of dry wood must be consumed.
This figure is certainly to be weighted since the fuel power of lignocellulose is 3 times lower than that of oil but remains aberrant in these times of energy saving
This figure is to be compared with an oil refinery that uses about 90 kg of crude oil to produce 1 ton of fuels and other products of our daily lives.
The goal will therefore be for the lignocellulose refinery completely green, to consume only 3x90kg or about 300 kg of dry biomass or one of its components to produce 1 ton of commercial products with high added value to be competitive first. with the oil refinery in terms of energy and make it totally green.
So if we want the 5 to 6 billion tons of agricultural and forest residues generated annually in the world, which are degraded into carbon dioxide and water without any economic and ecological contribution, replace, as they can, the 4 , 5 billion tons of oil that is consumed every year, so it will be necessary to think outside the box.
This is what I started to do more than twenty years ago to end today with the nice results that you will discover over the following lines.
CELLULOSE, HEMICELLULOSES, LIGNIN AND SILICA
These are the 4 main components whose structure is well known, with the exception of lignin, which for a long time was a real enigma.
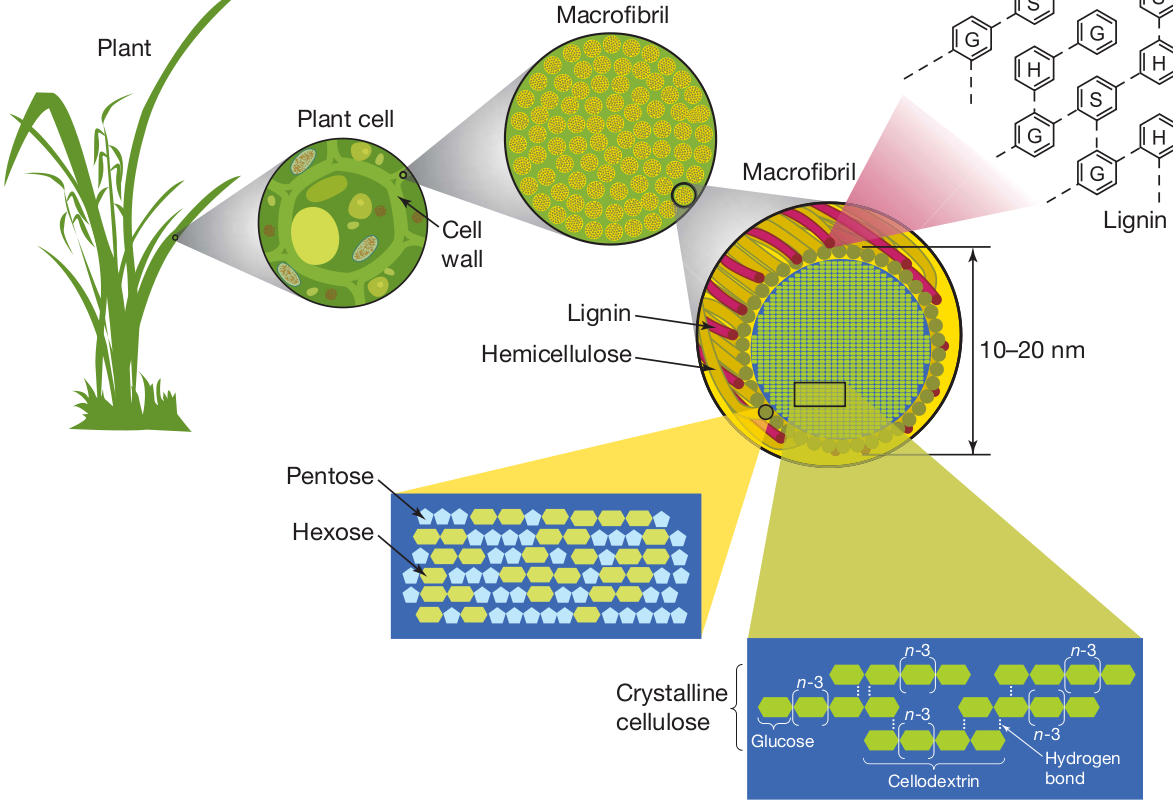
There are of course some molecules from specific secondary metabolisms up to a few%, characteristics of the raw material considered.
A little history shows that it was the chemist Anselme PAYEN, who, interested in the analysis of wood, showed in 1838 a substance that decomposed in glucose unit he called cellulose.
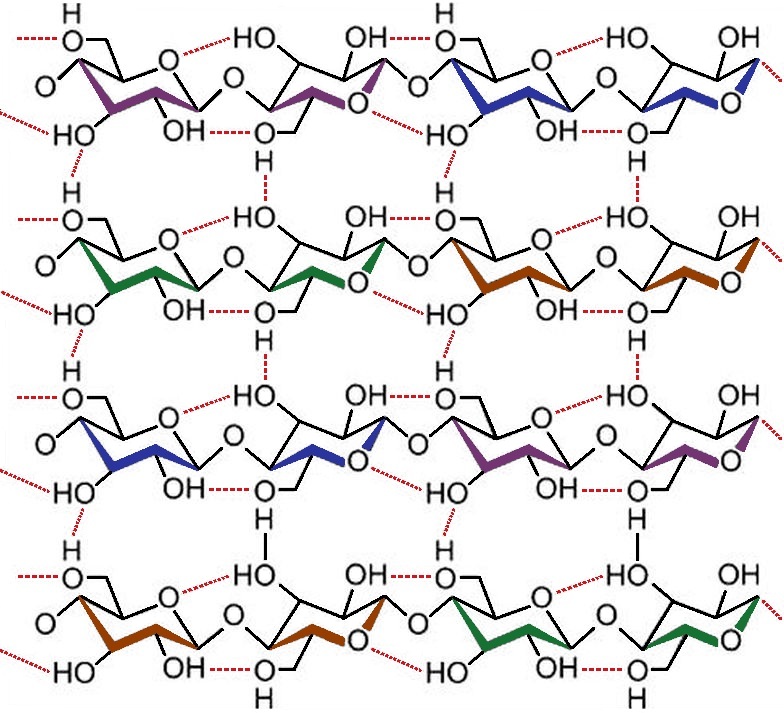
The following year, therefore, in 1839, he noticed the permanent presence of another substance around the cellulose fibers, which he described as an encrusting substance without going further in its definition. The term lignin appeared a few years later, in 1856, in the works of Franz Ferdinand SCHULTZE, confirming the observations and hypotheses of PAYEN.
Hemicelluloses were identified in the early twentieth century as polysaccharides found in terrestrial plants other than cellulose, starch and fructans.
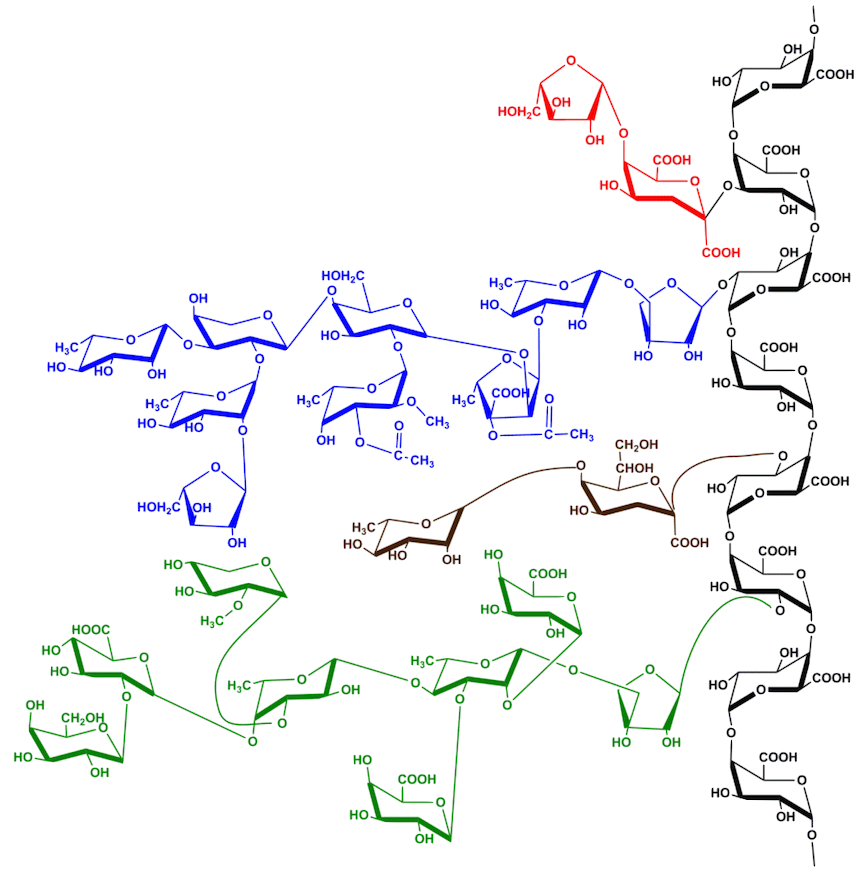
It must be said that the brutality of pulp extraction, which has dominated the world of lignocellulose until today, has an unfortunate tendency to demolish lignin and hemicelluloses. The resulting black liquor is heavily concentrated in organic and inorganic molecules whose complicated incineration can only recover a portion of energy and the chemicals used.
The existence of aromatic structures in the composition of lignins did not passionate then the scientific community. The black paper liquors, intimate mixtures of degraded lignins and sugars, oxidized sulfur and soda minerals were of course for many by their very unattractive character.
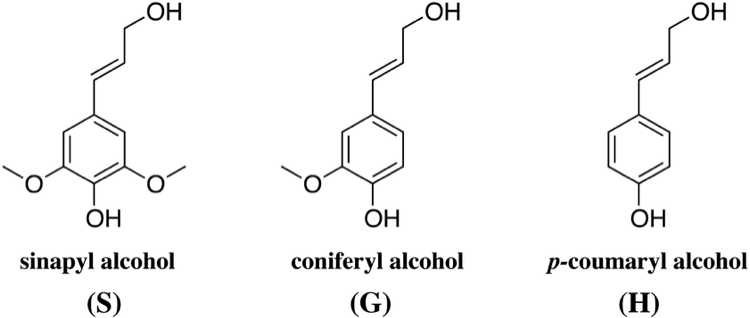
It was not until the end of the Second World War and the work of Bernard DAVIS that the so-called shikimic acid biosynthetic pathway explains how, via, inter alia, phenylalanine, glucose was passed to the following three allylic alcohols which constitute the monomers in apparently random distribution of lignin macromolecules.
ALDER proposed in the sixties a model that continues to appear in books and publications while it is far from accounting for the structure of lignin.
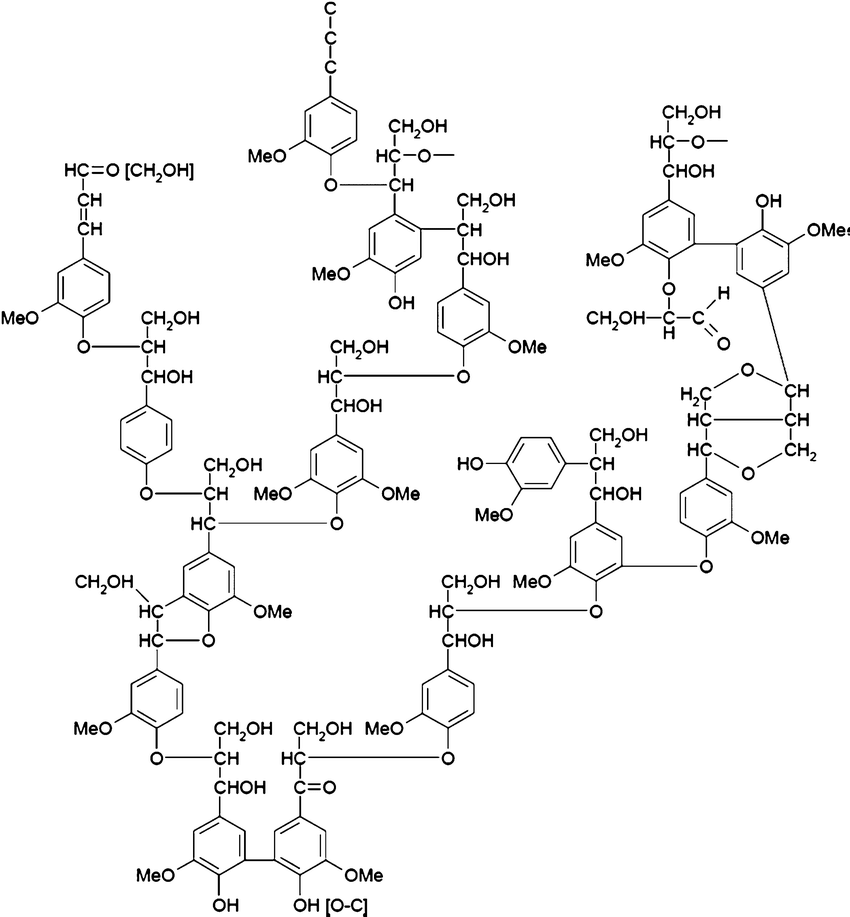
The arrangements of the polymer structure of lignin have been debated until the beginning of this century with essentially modeled structures far from physical realities.
Following my work on the separation of the main constituents of lignocellulose in weak organic acids such as formic acid and acetic acid, which I detail in the following paragraphs, I had the opportunity with my colleague, Professor JH BANOUB to propose for the first time in 2003 a simple linear oligomeric structure of low molecular weight, which caused a sensation at the time.
It is now widely recognized by the scientific community.
The absence of degradation of the natural macromolecule made it possible to eliminate any reference to a three-dimensional structure, and the extensive analyzes in mass spectrometry and NMR enabled us to establish, on solid experimental bases, the following generic formula:
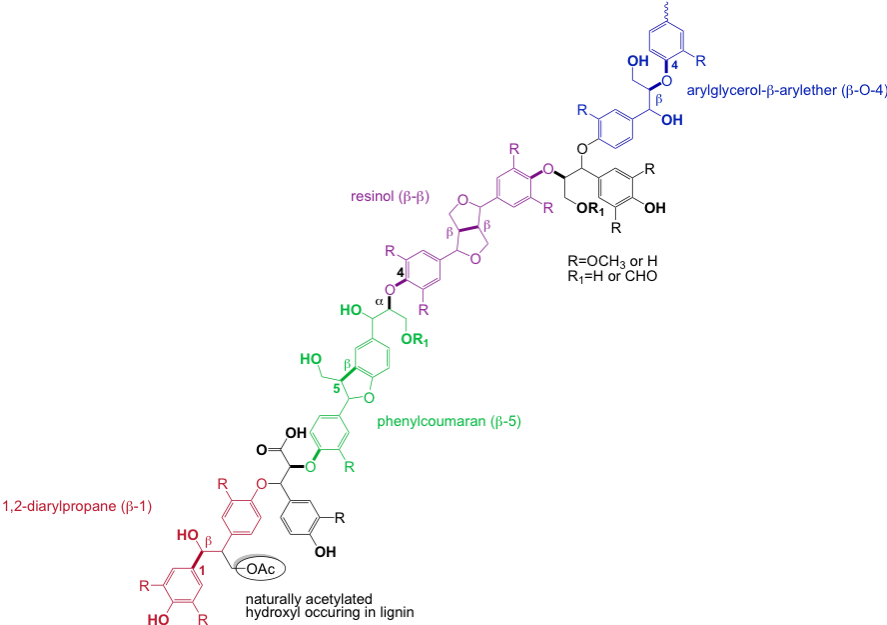
The partial esterification by the acetyl and formyl groups of the phenolic and hydroxymethyl groups that we have been the first to identify and quantify are of primary importance because of their impact on reactivity.
They simply depend on the proportions of acetic acid and formic acid present in the medium during the first stage of extraction as we will see later.
The total structural mastery we have about the lignin extracted by my technologies opens up spectacular ways of valorization which I will detail later.
As for silica, we confirmed its amorphous structure in the natural state in plants and can easily extract by solubilization / precipitation. Its numerous and lucrative applications are those of commercial precipitated silica. It will be an essential co-product for the refining of rice straw which contains more than 10% by weight.

ORGANOSOLV PROCESSES AND ASSOCIATED SOLVENTS
It is simply a matter of using solvents other than water to destructure lignocellulose, if possible, without using acid or basic hard media at high temperature and under pressure. The first works were conducted by Theodor KLEINERT in 1968 with methanol and ethanol.
About all the available solvents, hydrophilic or hydrophobic, were then tested with the primary objective, like the refining of oil, the separation without degradation of the lignocellulose components that we have just described.
Thus, over the years, for nearly a half century, we have encountered alcohols, inorganic and organic acids, ammonia and amines, ketones, ethers, various heterocycles that are more or less saturated, aromatic solvents, ionic solvents in a good tens of thousands of patents and scientific articles.
A hundred or so processes have reached the pilot stage and a dozen industrial stages that have not had a future. Only two to three unprofitable plants are trying to exist in the US and Brazil.
Nothing has seriously disturbed the secular extraction technologies of cellulose and therefore the paper industry to date.
I have long since, like other colleagues around the world, analyzed in detail all these technologies and the overall common conclusions are that at some point these technologies suffer from at least one of the crippling problems described below. which condemns their industrial development for economic and / or ecological reasons.
It is therefore in order of importance :
- The inability to carry out refining with complete absence of degradation of the components of lignocellulose,
- The toxicity, the cost of producing the solvent, its implementation and its complete recycling,
- The nature of the equipment and the associated construction and investment costs,
- The logistical and operational costs of the field or forest up to the competitive market with petroleum without subsidy of any kind.
In the end, among all the more or less reactive solvents of organic chemistry, after multiple laboratory tests with all the categories mentioned above, it is the first organic acids, namely acetic acid and formic acid that caught my attention.
They have never disappointed me for more than 20 years and will no doubt be, as you will see the key lignocellulosic biomass refineries that go, it is a certainty, gradually replace the oil refineries.
The two technologies that I developed are therefore ready for their industrial development with the process developed by the company CIMV with a TRL (Technology Readiness Level) of 8 and the very recent developed by BioEB with a TRL of 5/6.
These two technologies are at the same time scientific discoveries and innovations of industrial rupture at the forefront. To date, they have been the subject of 17 international patent families, 14 international trademarks and 17 scientific publications in the world’s leading scientific journals in their respective fields. All these references and published documents appear at the end of this presentation.
RAW MATERIALS
Lignocellulose today has little consideration from the major operators of energy production. They have logically, for the few biomass plants that operate around the world, used concepts to be urgently improved if we want, as the vast majority of countries, politicians and citizens alike, to do today. non-food biomass an effective reality of the energy mix of tomorrow.
It is therefore a mistake to correct in the future if we want to secure large scale supply chains essential to the energetic transition that concerns us all.
The equation is simple. Based on an oil price of $ 50 per barrel, which is the most reasonable today, it puts the ton of crude oil at $ 390. Under these conditions, knowing that the energy value of dry lignocellulosic biomass is 1/3 of that of oil, the price equivalent paid for dry lignocellulose, factory input by the industrial converting it, should therefore be 390 / 3 = 130 $ / T.
At this factory-paid price to the producer, the 4.5 billion tons of oil used in a year can be replaced tomorrow by existing lignocellulosic residues if the transformation technologies are as efficient as the current oil technologies.
Residues from the exploitation of food plants
Cereal straws

They are in the world an essential component of cereal production. According to FAO, the world’s cereal production in 2016 was 2 million tons, so at least as many straws.
At the moment most of them go up in smoke or are degraded in carbon dioxide and water without any profit for the grounds and the farmers who work them. They constitute a resource of nearly 2 billion tons immediately available in most countries of the world, so well they are remunerated at fair value.
Bagasses of sugar cane and sweet sorghum

As for cereals, bagasse, the lignocellulosic part of sugarcane and sweet sorghum, accounts for about 50% of the dry matter, as much as sugar. The annual production of sugar last year was 187 million tons. It is recognized today that with an optimization of the conversion of bagasse by cogeneration into steam and electricity, a good third or more can be exported on the network, the rest feeding the plant. Vegetal refining can, without diminishing the achievements, seriously increase its efficiency and profitability.
Logging residues: hardwoods
Hardwoods

It is in the northern hemisphere that hardwoods, often left on the ground during the exploitation of conifers, constitute an enormous resource readily available.
Compte-tenu de leur croissance plus rapide et des chiffres d’exploitation des bois ronds qui dépassent largement le milliard de tonnes, la voie est ouverte à des quantités équivalentes pour le raffinage végétal et donc à la production simultanée de produits à haute valeur ajoutée et d’électricité.
Les palmiers à huile

It is a denied sector with many objective reasons for accelerated deforestation but currently produces 65 million tons of oil from fruit and kernel for biodiesel and the food industry. If we add to this that for a ton of oil produced, it is nearly 6 T of lignocellulosic waste that is generated and which therefore decompose under somewhat questionable conditions, it becomes downright frightening.
Since these residues are fully exploitable, just like wood and straw because of their identical composition, nearly 400 million tons of lignocellulose are available to enter the virtuous circle of vegetal refining. The industry then takes another look.
The recent report on the subject of IUCN (International Union for Conservation of Nature – https://doi.org/10.2305/IUCN.CH.2018.11.fr) which cannot be suspected of collusion with producers, confirms the about and is quite instructive.
Specific lignocellulosic productions

It is a way to consider on land that cultural practices and / or industrial pollution have made unsuitable for food production. For example, miscanthus can play a very interesting role because spaces will not fail.
My remarks are not exhaustive given the huge annual global production of terrestrial biomass from atmospheric CO2, which happily exceeds 200 billion tons.
Photosynthesis produces annually more than we consume annually. For two centuries, oil and coal could well have remained underground if biomass had the same time and investments.
This is a crazy challenge that I attacked a little over twenty years ago and you will see all its interest through the creation of CIMV and BioEB which you can see their progress by clicking on their link.
The bibliographical references which follow give the elements of access to the non-confidential details of the works carried out under my Direction for more than 20 years on my concepts of vegetable refining in particular the patents which were deposited in all property by CIMV and by myself exploited under exclusive license by BioEB.